ASCIA Action, First Aid, Management, Transfer, Travel and Treatment Plans
- ASCIA Action Plans for Anaphylaxis (RED)
- ASCIA Action Plan for Drug (Medication) Allergy
- ASCIA Record for Drug (Medication) Allergy
- ASCIA Action Plan for Allergic Reactions (GREEN)
- ASCIA First Aid Plans for Anaphylaxis (ORANGE)
- ASCIA Travel Plan
- ASCIA Action Plan and Management Plan for Eczema
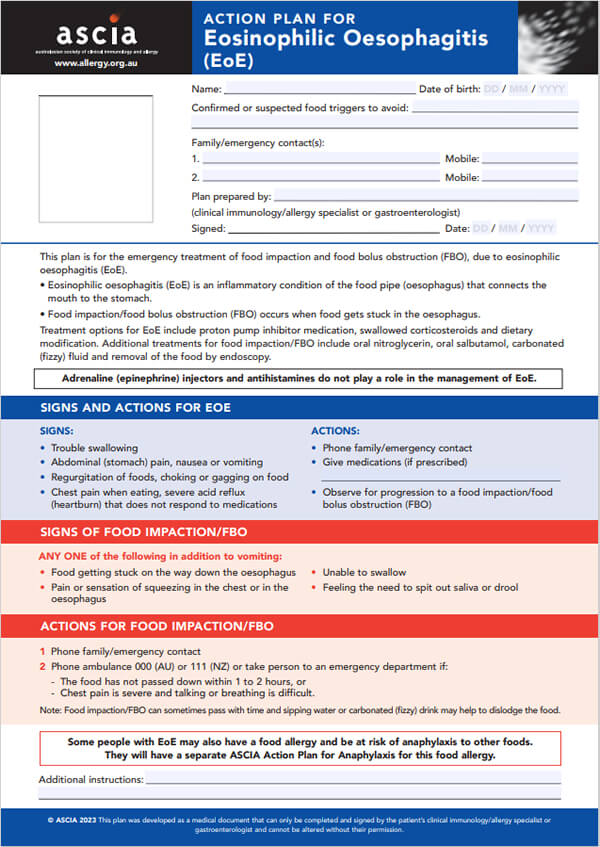
- ASCIA Action Plan for EoE
- ASCIA Action Plan for FPIES
- ASCIA Management Plan for Delayed Allergic Reactions to Foods
- ASCIA Immunodeficiency Plans
- ASCIA Management Plan for HAE
- ASCIA Treatment Plan for Allergic Rhinitis
- ASCIA Treatment Plans for Immunotherapy
ASCIA Action Plans for Anaphylaxis
ASCIA Action Plans for Anaphylaxis are emergency response plans for severe allergic reactions (anaphylaxis). Major updates were made in the 2023 versions, and only minor updates have been made to the 2025 versions. Prior versions (2023) will still be valid for use in 2025.
For further information about ASCIA Action Plans go to www.allergy.org.au/hp/anaphylaxis/action-plans-for-allergic-reactions-faq
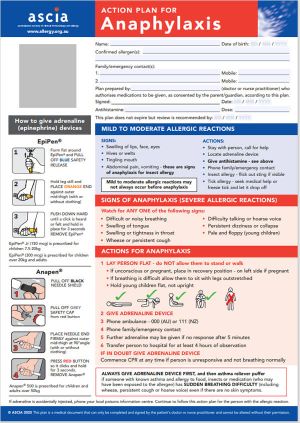
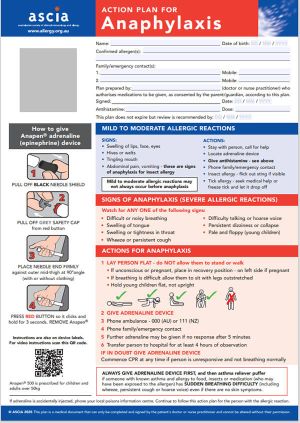
ASCIA Action Plan for Anaphylaxis (RED) 2025
The general version of this plan is for people with allergies who have been prescribed either brand of adrenaline device.
Device specific versions are available below.
These plans are not for allergic rhinitis (hay fever) due to pollen, dust mite, animals or other inhaled allergens.

ASCIA Action Plan for Drug (Medication) Allergy (GREEN) 2025
This plan is provided to people with drug (medication) allergies who are not usually prescribed an adrenaline device, because accidental exposure to drugs can mostly be avoided. They are usually recommended to wear medical identification, to avoid exposure to drugs used in medical procedures.
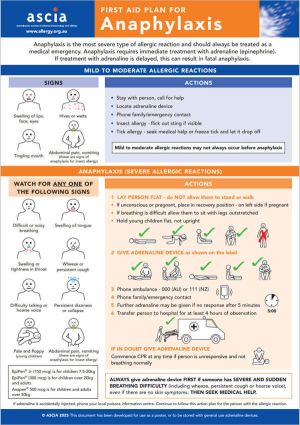
ASCIA First Aid Plans for Anaphylaxis (ORANGE)
This plan can be used as a poster or stored with general use adrenaline devices.
A version of this plan for use by crew on aircraft is available upon request.
For translated versions of the ASCIA First Aid for Anaphylaxis go to www.allergy.org.au/hp/anaphylaxis#ta5
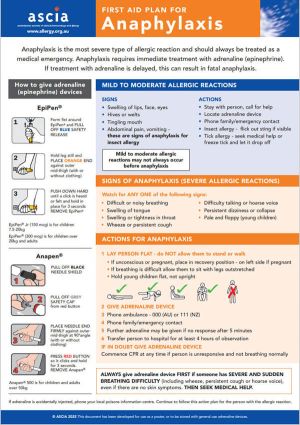
ASCIA First Aid Plan for Anaphylaxis (ORANGE) 2025
The general version of this plan can be used as a poster or stored with either brand of general use adrenaline devices.
Device specific versions are available below.
ASCIA First Aid Plan for Anaphylaxis (ORANGE) 2025 Pictorial Poster
This plan can be used as a poster or stored with general use adrenaline devices.
ASCIA Travel Plan for people at risk of anaphylaxis 2023
This plan should be used with an ASCIA Action Plan for Anaphylaxis.

ASCIA Management Plan for Delayed Allergic Reactions to Foods
![]() ASCIA Management Plan for Delayed Allergic Reactions to Foods 2023
ASCIA Management Plan for Delayed Allergic Reactions to Foods 2023

ASCIA Treatment Summary for Patients on Immunoglobulin Therapy
![]() ASCIA Treatment Summary Immunoglobulin Therapy 2024118.04 KB
ASCIA Treatment Summary Immunoglobulin Therapy 2024118.04 KB
Content updated August 2024
ASCIA Action Plans,
Treatment Plans and
Checklists
- Health Professionals
- ASCIA Action Plans, First Aid Plans,
Treatment Plans and Checklists - Anaphylaxis Resources
- Health Professionals e-training
- Position Papers/Guidelines
- Allergic rhinitis | Sinusitis | Asthma
- Drug allergy
- Podcast - Antibiotics and Allergy
- A career in Clinical Immunology
and Allergy - Awards/Grants
- Research Studies
- ASCIA ID Register
- ASCIA Action Plans, First Aid Plans,